Watch Plating 101 Primer
One of the most frustrating issues for an owner of a vintage, or heirloom watch in need of replating, is sorting through all the unfamiliar technical terms that are used to describe plating thickness, quality standards and best practices.
Navigating through all of this data can be very frustrating even for the most knowledgable consumer. This frustration is understandable. The electroplating process is complicated. Explaining it in basic terms without oversimplification is also difficult. In some cases, well intended explanations offered to the public have led to a huge amount of misinformation, amusing urban myths, and unnecessary technical biases within the jewelry trade, and the watch repair field.
Sometimes the time the prized watch in need of replating, will end up back in the jewelry box, or dresser drawer. For those who enjoy the beauty and pleasure of wearing a distinctive vintage watch, this is a shame.
So instead of just storing that watch away again, lets look at some of the key technical terms, and basic concepts that will help you to know how watch replating works.
Explaining all of the important details without compromising technical correctness is our goal here. At the conclusion of this article, you can also refer to the frequently asked question
section on watch replating. That section will help you put the technical information from this article into a "real world" context.
Before we get into the technical issues, here's a little background about electroplating services in general. First, you should only trust a qualified electroplater who has an appropriate level of experience (at least 20 years).
Unlike any other jewelry object, watchcase, and band replating are highly specialized, and are some of the most difficult decorative items to replate correctly. With rare exceptions most jewelers, jewelry repair shops and hobby electroplaters are not properly equipped to replate a watch to the rigorous standards that meet, or exceed the original specifications of the manufacturer. From a durability and wear standpoint, high quality watches rely on a heavier layer of gold than do most electroplated jewelry. That type of heavy gold electroplating requires exceptional knowledge of electrochemical principles, specialized plating equipment, rigid compliance to proper technical processes, and careful quality control during each step of the electroplating process.
As a result, one of the biggest challenges will be finding a qualified electroplating resource! While there are many electroplating shops in the US, their operations are geared to plate objects on a mass production scale, or they are focused on only specialized industrial-based electroplating applications. They view custom watch replating as outside of the scope and scale of their operations.
There are a number of custom retail plating operations. Some of them are qualified and knowledgeable about jewelry replating. However, that does not mean that they are interested, or experienced specifically in the unique methods needed to refinish, and replate a watch.
First a little background on the electroplating process and its basic concepts. Electroplating can be defined as the depositing of a very thin layer of metal "electrolytically" to a base metal to enhance or change its appearance. Atoms are bonded together in the same way they are bonded in a solid piece of metal.
Electroplating is done in a liquid solution called an electrolyte (fig.a), also known as a "plating bath". The plating bath is a specially designed chemical bath that has the desired metal (i.e. silver, gold) dissolved as microscopic atoms (positive charged ions) suspended in solution. The plating bath solution serves as a conductive medium and utilizes a low d.c. voltage (direct current). The object that is to be plated is submerged into the plating bath and a low voltage d. c. current is applied to the bath. Generally located at the center of the plating bath, the object that is to be plated acts as a negatively charged cathode (fig. b). The positively charged anodes (fig. c) that will complete the d.c. circuit are carefully positioned at the edges of the plating tank. A power source known as a rectifier (fig. d.) is used to convert a.c. power to a carefully regulated low voltage d.c. current.

The resulting circuit channels the electrons into a path from the rectifier to the cathode (object being plated), through the plating bath to the anode (positively charged) and back to the rectifier. Since electrical current flows from positive to negative, the positively charged ions at the anodes flow through the plating bath's metal electrolyte toward the negatively charged cathode. This movement causes the metal ions in the bath to migrate toward extra electrons located at the cathode's surface outer layer. By means of electrolysis, the metal ions are taken out of solution and are deposited as a thin layer onto the surface of the object.
This process is called electrodeposition. Theoretically, the thickness of the electroplated layer deposited on the object is determined by the time of plating, and the amount of available metal ions in the bath relative to current density. The longer the object remains in the d.c. activated plating bath, the thicker the electroplated layer will become.
Bear in mind that this brief explanation of the electroplating process is very basic. The actual processes, and specialized equipment as they relate to watch replating are considerably more complex. There are a large number of technical parameters each having wide variations that must be correctly controlled and monitored in order for the electroplating process to work properly.
Now that we have a basic introduction to the electroplating process, lets examine the key issues that contribute to successful watch replating. There are 7 basic factors that will impact the overall beauty, luster and long term durability of your replated watch. In reality there are a great deal more than seven, but they have more to do with procedural steps during the electroplating process and are outside of the scope of this article.
The seven factors are
:
1.
Thickness
of the goldplated layer as it relates to durability, discoloring, and the overall average gold thickness in the recessed areas of the watch case.
2.
Hardness
of the gold plated layer as it relates to scratching, incidental abrasion and resistance to chipping.
3.
Adhesion
of the gold plated layer to the surface of the watchcase.
4.
Underplating
of the goldplated layer with copper and nickel to improve tarnish resistance.
5.
Porosity
and its elimination within the gold plated layer as it relates to resistance to oxidation and diffusion.
6.
Color and tint
of the goldplated finish relative to aesthetics and the original manufacturers specifications.
7.
Surface finish
as it relates to cleaning and repolishing prior to the final electroplating process
Lets go through each factor and highlight the most important points to understand how they contribute to the success of watch replating.
factor 1 -
gold thickness:
For the finish and luster of a replated watch to last at least 7-10 years or more of regular daily use, you will need a respectable thickness of plated gold. From a corrosion resistant standpoint it will be just as important to have the unseen recessed areas (i.e. threaded areas, lugs, crystal edges) of the watch case to also have an adequate thickness of gold plating. Before we get into that level of detail, lets take a moment and learn about how goldplated thickness is measured within the jewelry industry.
As a starting point it will be necessary to define the "unit of measurement" that describe gold plated layer thicknesses. The most common unit of measurement in the US is the "microinch". In layman's terms, a microinch is actually the same as one millionth of an inch (see chart a).
For Europe and some U. S. industries (i.e. jewelry trade), the unit of measurement is often expressed in "microns." As an example, the edge thickness of a US dime coin is 1250 microns or 1.25 millimeters.

Plating thicknesses for precious metal objects and related trade is regulated by specific rules that are administered by the US Federal Trade Commission. If you are interested in more detail, additional information can be found in the Code of Federal Regulations, Title 16, Commercial practices Part 23 January 1999. Precious metals under FTC regulations include gold, silver, and the platinum metals group. FTC requirements for plating thickness are expressed in both microinches and microns (see chart b).
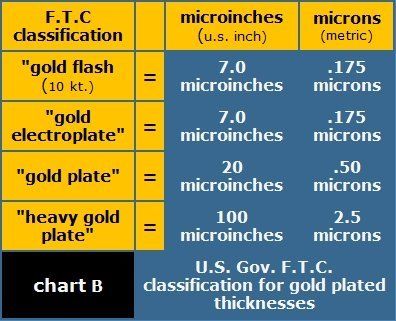
For an even clearer comparison, chart c describes the average thickness in microinches and microns of some common objects most all of us are familiar with. As you can see most electroplated gold finishes are actually quite thin by comparison.

After comparing chart b and chart c, the first thing one would ask is, how can any gold electroplating as thin as what is designated by the FTC regulations last very long? For watches you would be right to ask that question.
However, bear in mind that in the decorative world of gold watch replating 5-7 microns is quite heavy! Combined with the addition of "alloys" to harden the gold plating, this thickness of electroplating is more than adequate for most replated watches.
Some electroplaters might tell you that 10-20 microns is the correct thickness for a real authentic "Swiss quality" watch replate. If you are replating with a layer of 99.9% fine pure gold with no hardening alloy, that viewpoint might be right. Depending on the watch case design, you would need this heavier layer of fine gold because the softness of the plated gold would start to rub-off the watch's surface through normal wear.
However, with today's goldplating formulas, extremely hard gold finishes with excellent resistance to scratching and abrasion are available. As a result, beyond a certain threshold it is not necessary or advantageous from a durability and "rub-off" standpoint to replate watches above 7 microns.
From a technical perspective, proper thickness of the gold plated layer also helps minimize the impact of "porosity" within the surface structure of the replated watch. We will talk in more detail about porosity and its relationship to premature surface discoloring later in the article.
It is just not entirely true that plating thickness on the high wear areas of the watch determines the durability of the replated watch's finish. Thickness is important, but the overall "average thickness" of the goldplating over the entire surface area of the watch is just as important.
The "physics" of the electroplating process by its very nature has a tendency to deposit the gold on any object unevenly. This is especially true for the intricate recessed areas and contours within watch case design. Prominent edges and significant decorative surface (the exposed areas around the watch crystal and lugs) will receive a thicker plating than the recessed areas within the watch case.
To prevent any surface oxidation and discoloring from propagating from the unseen secondary areas of the watch case, it is very important that a replated watch have adequate gold thickness on every portion of its surface area.
A goldplated layer with at least 3-4 microns in the recessed non-show areas of the watch case is quite effective. This level of gold thickness will eliminate corrosion from starting within the watch. This is especially true for the threaded areas of the case around the removable back cover of the watch and the lugs that secure the watch band. Those portions of the watch's surface are in constant incidental contact with the oils of the skin, chemicals, and perfumes.
factor 2 -
Hardness of the Gold Plated Layer:
Its not enough to have a thick goldplated layer if it is easily rubbed off from casual contact with clothing, skin and other surfaces. If the gold plating is soft it will be easily worn off over a short period of time. This will be especially noticeable around the high points of the watch and the primary edges near the lugs. Surface hardness and toughness of the final gold plated layer is extremely important for the watch to be resistant to scratches and incidental abrasion.
Unlike goldplating thickness, hardness for goldplating is no longer regulated or specified by the FTC (as of august 1999). The manufacturers within the watch industry have technical guidelines for recommended hardness that are still utilized on newly manufactured goldplated watches.
Commercial electroplating shops who goldplate objects such as plumbing fixtures will defer to Military Specifications for finish hardness specifically MIL-G-45204C. In that specification, hardness for goldplating is referred to as "grade" (see chart e). For our replating applications, we will rely on this aspect of this specification for determining the standards for the different levels of hardness available for gold plating of watches.

The standard for measuring the hardness of gold plating is expressed as "knoop hardness". Knoop hardness is calculated by measuring the indentation produced by an elongated four-sided pyramid shaped diamond point. Pressed into an electroplated surface under loads (usually less than 1 kilogram-force) the indenter creates a 4 sided impression. Hardness numbers are cited relative to specific load values that were used in the test.
A knoop hardness value for a replated gold watch should be rated at least 110-130 range (grade b).
This hardness value will provide good scratch resistance and long term finish durability for watches. You could designate a harder finish, however the tendency toward embrittlement of the goldplating can become an issue. In some cases this could lead to minor chipping of the heavy goldplated layer, particularly on the sharp edges and prominent design contours of the watch.
factor 3 -
Adhesion of the gold plated layer:
This is getting into territory that is harder to control, and relies heavily on the best practices, judgment and skill of the electroplater. The purpose of including this information in our discussion is to raise the readers overall awareness.
Watch cases (except for solid gold types) are made out of brass or
some type of copper/bronze alloy.
Some watch cases are nickel. Brass and its alloys have a tendency to oxide and tarnish very quickly when exposed to normal environmental conditions. Protecting it from discoloring is one of the reasons the watch case was goldplated in the first place.
Contaminants from oxides formed on the brass watch case can prevent good adhesion of the goldplated layer. Before a brass watch is replated, all of the worn out gold plating and surface oxidation must be removed. The brass surface must be carefully refinished and repolished. Prior to the plating process the surface must be free of any oxides or contaminants. The cleaner the surface the better the adhesion. One of the things that is often overlooked during this phase is the
microscopically imbedded
contaminants in the watch's outermost surface layers. This normally occurs during the final polishing steps and cleaning just prior to the plating process.
Any contaminant in the form of abrasive grit, polishing compound, soap residue, or oxides on the brass watch case can adversely affect surface adhesion. By adhering to careful cleaning processes, the electroplater can overcome most adhesion issues by submerging the watch case in a light acid rinse to remove any oxide films and to activate it's surface area. From this point great care must be taken to avoid any recontamination from quick forming oxides, fingerprints or oils during handling prior to the final plating process.
Lack of good adhesion is one factor in replating that can often lead to surface porosity or blistering in the goldplated layer.
factor 3 -
Adhesion of the gold plated layer:
This is getting into territory that is harder to control, and relies heavily on the best practices, judgment and skill of the electroplater. The purpose of including this information in our discussion is to raise the readers overall awareness.
Watch cases (except for solid gold types) are made out of brass or
some type of copper/bronze alloy.
Some watch cases are nickel. Brass and its alloys have a tendency to oxide and tarnish very quickly when exposed to normal environmental conditions. Protecting it from discoloring is one of the reasons the watch case was goldplated in the first place.
Contaminants from oxides formed on the brass watch case can prevent good adhesion of the goldplated layer. Before a brass watch is replated, all of the worn out gold plating and surface oxidation must be removed. The brass surface must be carefully refinished and repolished. Prior to the plating process the surface must be free of any oxides or contaminants. The cleaner the surface the better the adhesion. One of the things that is often overlooked during this phase is the
microscopically imbedded
contaminants in the watch's outermost surface layers. This normally occurs during the final polishing steps and cleaning just prior to the plating process.
Any contaminant in the form of abrasive grit, polishing compound, soap residue, or oxides on the brass watch case can adversely affect surface adhesion. By adhering to careful cleaning processes, the electroplater can overcome most adhesion issues by submerging the watch case in a light acid rinse to remove any oxide films and to activate it's surface area. From this point great care must be taken to avoid any recontamination from quick forming oxides, fingerprints or oils during handling prior to the final plating process.
Lack of good adhesion is one factor in replating that can often lead to surface porosity or blistering in the goldplated layer.
factor 4 -
Underplating:
Underplating is closely related to
factor 3
with regard to improving surface adhesion of goldplating. As its name implies, underplating is a series of plating processes that are used to prepare a watch for it's final plating in gold. The intent of underplating is to improve the tarnish resistance of your replated gold watch.
After the watch as been repolished, cleaned and rinsed, it is electroplated with a layer of bright copper. Electroplated copper readily adheres to the brass watch case and provides a good intermediate layer to help prevent porosity and the formation of oxides. For badly pitted and gouged watch cases, sufficient layers of copper can be plated onto the surface to fill and smooth out surface irregularities. After copper is plated onto the watch it is prepared for the next step.
Depending on the manufacturer, many medium and high priced vintage watches were also underplated with a layer of bright nickel prior to final goldplating. Bright nickel has many desirable characteristics that help improve the luster and durability of your replated watch. The most noteworthy advantage of nickel underplating is its ability to prevent corrosion from the brass watch case from reaching the goldplated layer. If there is any porosity in the goldplated layer, the nickel layer will prevent any surface contaminants from corroding the brass watch case; which in time would discolor the goldplated layer. Finally, a layer of nickel plating can help brighten and smooth out any minor surface irregularities on the watch case.
factor 5 -
Porosity:
It would be easy to write a whole chapter on the many important technical relationships that can lead to porosity (i.e. transverse pores, bulk porosity) in goldplated finishes.
When electroplating begins forming the first layer of gold onto the watch case, the gold is gradually constructed at a molecular level in a "lattice type" layer structure. The longer the watch is exposed to the plating bath the thicker and denser the gold lattice layers will be.
If there is any contamination on the surface of the watch case or uneven roughness (i.e. scratches) in the finish, the molecular lattice structure of the gold can be interrupted and a microscopic "pore" can form and begin growing at the contact point. If this and other factors (i.e. plating baths, power supply) in the electroplating process are not tightly controlled, porosity resembling layers of microscopic swiss cheese will occur throughout the goldplated layer.
Unfortunately, porosity can allow the brass watch case to slowly corrode as surface contaminants migrate through the tiny pores in the gold layer. Eventually this corrosion will travel back to the surface of the goldplated finish of the watch and cause it to change color. This is one of many reasons why the gold finishes of replated watches have such a bad reputation for not lasting very long. Therefore, it is vital that all porosity be minimized or completely eliminated during the gold electroplating process.
There are a few ways to achieve this. First, you should keep in mind that there appears to be an important relationship between gold thickness and porosity formation. Simply stated, porosity decreases as the gold layer becomes thicker. As the goldlayer is built up, new layers of gold bridge over and "seal" any pores that were formed during the beginning stages of the electroplating process. Goldplating in sufficient thicknesses combined with proper electroplating processes can virtually eliminate any chance of surface contaminants from migrating into the brass watch case. This is why a heavier layer of goldplating should be utilized.
It is important that the plating process utilizes some type of protective underplating process. As mentioned earlier, bright nickel can be utilized as an underplating to prevent any contaminants from traveling to or from the brass watch case and eventually discoloring the goldplated layer.
factor 6 - Color and tint
:
Matching the original color of your vintage watch is important to assure an authentic and aesthetically pleasing look that best duplicates its original factory finish!
Contrary to popular belief the color of the gold plating has very little direct relationship to the purity rating of the gold being used. Different goldplated colors are achieved by adding small amounts of alloy metals to change the "color and tint" of the final gold plated finish.
As an example, if your watch had a designation of "14 karat gold plating", that color was probably achieved by adding small amounts of either nickel, cobalt or palladium (or combinations of all of them) to "pale it down" from the distinctive yellow-orange color of pure 24 karat gold. Other techniques such as changing the temperature of the plating bath, adjusting the d.c. voltage, or simple agitation of the watch case in the plating bath, can allow the color of the plated gold to be modified even more.The goldplated layer might still be 90% pure gold, but its color would look like it is a 14 karat gold finish (actual 14 karat gold is 58% pure relative to 24karat which is 99.99% gold). Therefore you should know that when you designate a 14kt or 18kt goldplated finish for your watch, the final finish color may have no direct relationship to the actual gold purity deposited on the watch.
As a sidebar, with the exception of large scale or high volume plating jobs, the actual cost differences between 14kt color goldplated and 24 karat color goldplated items are insignificant. In actuality, yellow 14 karat hard gold platings for watches (5-7 microns) are more difficult to plate (i.e. embrittlement from internal stressing,) but are far more durable than non-alloyed pure 24 karat color goldplated finishes.
With this family of 14 karat color goldplating finishes it is quite easy to duplicate many of the "yellow hamilton" gold colors used in the fifties and sixties on mid to high priced watches. In addition, they have great color, excellent scratch resistance, look less garish, or fake than the 22kt or 24kt color goldplated finishes. If your looking for more exotic colors for your watch, green gold ,rose gold, gray gold and purple gold plated finishes are also available. These types of gold plated finishes have been alloyed with other metals to achieve their color. They still appear gold, but have a "tint" to their finish from the alloy that was added to the plating bath.
Beware, some of the highly advertised "secret formula" 99.9% pure 24 karat goldplated finishes that are being offered. They appear bluish-yellow (cobalt alloyed) in color and are far from the desirable "warm yellow" color of the original watch finish. That does not mean that their finish is not durable or executed well, but they will have an undesireable color.
factor 7 - Surface finish
:
One of the most important and easiest factors to control in watch replating is the quality and brightness of the watch case's surface finish. Without special underplating, the electroplating process does not hide pre-existing scratches or pits. If anything the plating process will make a bad surface finish look even worse.
With this in mind it is important to spend extra time in the refinishing phase to remove deep scratches, fill in all surface pits and to rebuild worn design edges. This part of the process can actually take more time than the electroplating, especially if the watch is older and the contours of the watch case are badly worn.
Sometimes the deep pits and surface gouges can be gradually filled with a suitale metal during the underplating process. Other times it is necessary to melt a special gold or silver solder directly onto the bare brass watch case to fill any deep surface imperfections. This can be time consuming and requires a great deal of skill on the part of the refinisher. All prominent design edges must be redefined and polished. In addition all minor chips or gouges around the lugs that hold the watch band must be carefully removed.
It is better to fill deep scratches and surface imperfections than to try removing them with abrasive sanding. Too much sanding can ruin the watch case by making it too thin. This is especially critical for the "design contour" areas of the watch case that define its decorative beauty or contribute to its structural integrity (i.e. lugs, bezel around watch crystal).
After the major surface imperfections are removed, final polishing of the watch case to remove all other fine scratches are necessary. At this point all satin finishes should be uniform in appearance, and all shiny surfaces should be mirror bright and free of any surface irregularities. Finally, the watch is now ready to be replated.
This may seem like an extraordinary effort and it is. One could describe this process more accurately as "watch restoration" rather than just repolishing and watch replating. In some cases, if done correctly these important steps can greatly increase the monetary and intrinsic value of your vintage watch.
From a technical point of view, based on the 7 factors included in this article, we can now summarize the perfect watch specification for replating a watch with gold:
a. Removal of all of the old plating, scratches and surface imperfections including all pits.
b. Refinish and repolish of entire watch surface.
c. Underplated with a layer of bright copper.
d. Underplated with a layer of bright nickel.
e. Gold plated thickness of 5-7 microns on primary surface, 3-4 microns of gold in all recessed areas of the watch case.
f. Gold hardness of 110 knoop to 130 knoop (based on specification MIL-G-45204C grade b).
g. Color of gold plating to match original manufacturer specifications.
It is important to keep in mind that due to the different watch case designs, surface contours, case condition and other factors, each watch in need of goldplating presents its own unique requirements for refinishing and replating. The ultimate price determinant for any watch replating job will be based on the level of refinishing you decide to have done.
Lets talk about pricing for a replated watch. One of the dead giveaways of an inferior quality replate can be detected in the initial price estimate. If you are quoted 150 dollars or less to replate your watch, you can assume that the price does not include the removal of the mechanical parts from the watch casing. It will also not include the careful refinishing and surface restoration that will be required prior to replating. In some case it may not include even the basic repolishing! Make sure you ask enough questions to ensure that you know what you are getting for your money.
Some watch replaters will tell you that they meet all of the government standards for heavy goldplating (2.5 microns), including military standards for gold plating; and they probably do. But based on the 7 factors reviewed earlier that is not enough for the type of watch replating that guarantees a long lasting finish.
The standard end of this price range is based on a watch in reasonably good shape, with no deep surface scratches or damaged lugs. This price would include a top quality repolishing operation and replating in gold with a thickness of 5-7 microns.
If the case and/or band are in very poor shape with deep imperfections or damage required filling and/or replacement then extra cost may apply. We will contact you before proceeding if this applies.
Final Thoughts and Views
So there you have it, everything you need to know about basic watch replating. This information should be more than enough to help you understand the importance of a qualified resource to refinish and replate your watch.
For additional information regarding electroplating and metal refinishing, be sure to visit our
FAQ section located on this site. If we can assist you in your custom plating and metal refinishing needs, please contact us at the e-mail address below, or phone us to discuss the specific services you may require.
All articles copyright of The Time Preserve. Reproduction of this article in any form without expressed permission is not allowed. All rights reserved. Thank-you!
















